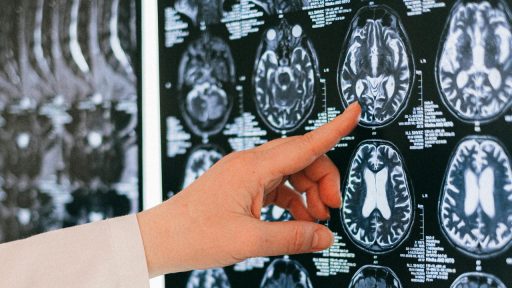
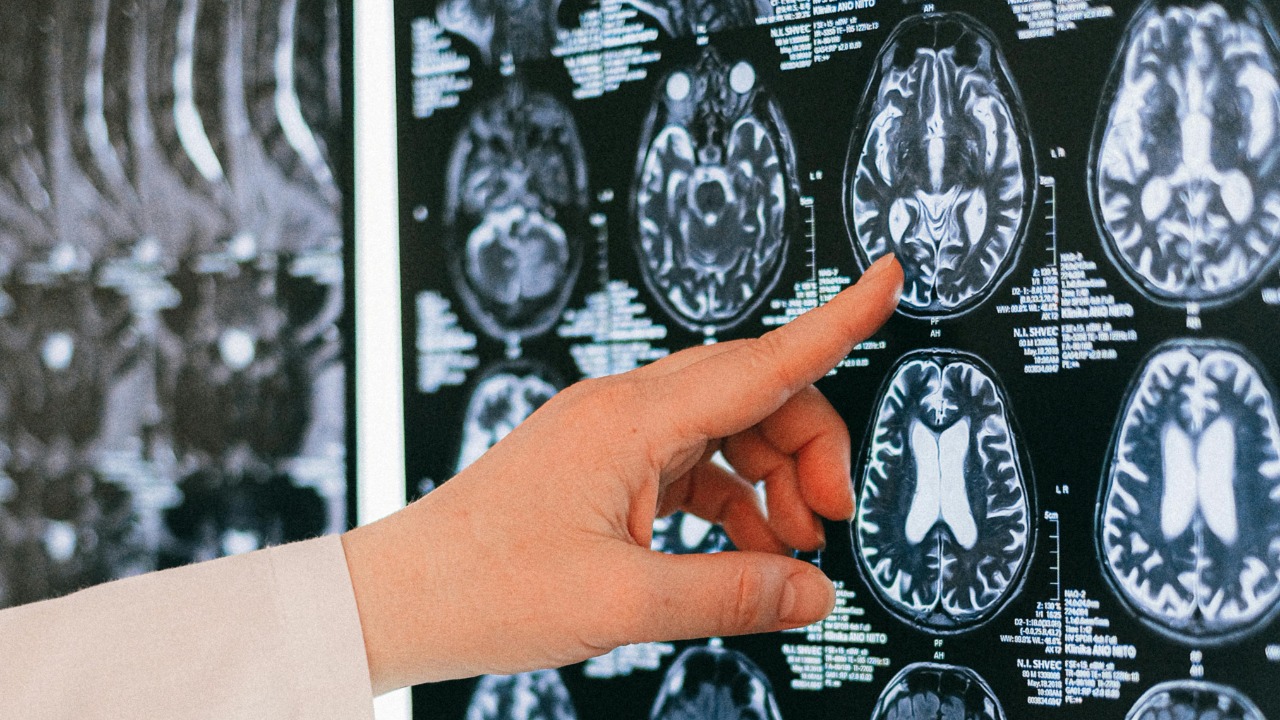
Alzheimer’s disease becomes dramatically more common with age, yet the biological trigger behind that steep climb has remained stubbornly unclear. A new wave of research points to a subtle malfunction inside the aging brain that scrambles how neurons clear waste, handle proteins, and communicate with each other. Together, these findings suggest that a specific kind of “brain glitch” may be what turns ordinary aging into a high-stakes risk for dementia.
Rather than focusing only on the plaques and tangles that show up late in the disease, scientists are tracking earlier changes in the brain’s drainage systems, protein quality control, and support cells. The emerging picture is that age gradually sabotages these hidden processes, and once enough of them fail, the brain’s defenses against Alzheimer’s start to collapse.
When the brain’s drains start to clog
One of the clearest clues comes from the brain’s own plumbing. Researchers have found that tiny fluid-filled channels that run alongside blood vessels, known as perivascular spaces, can become enlarged and clogged in people at higher risk of Alzheimer. In work highlighted in Jan, Researchers discovered that these “drains” show signs of blockage early, before obvious memory loss, suggesting that waste products and inflammatory molecules may be backing up long before a diagnosis.
The same study reported that Enlarged perivascular spaces were linked to four of the seven biochemical measurements taken in the participants’ brains, a pattern that ties structural changes in these channels to specific shifts in brain chemistry. That connection, detailed in Jan as Enlarged spaces, suggests that clogged drains are not just a bystander but part of the mechanism that pushes an aging brain toward disease. If clinicians can spot these changes on routine scans, they may gain a warning signal years before lasting brain damage occurs.
A hidden process that scrambles synapses
Drainage is only one part of the story. A separate line of work, summarized by Ivan Farkas in Feb, points to a hidden brain process that affects how neurons maintain their synapses, the junctions that carry signals. The report on Hidden Brain Process describes how age-related shifts in cellular housekeeping can quietly erode synaptic function long before neurons die. As this maintenance system falters, synapses become less responsive and then disconnect, a pattern that matches the early memory slips families often notice first.
In that account, the phrase Hidden Brain Process May Help Explain Why Alzheimer and Risk Rises With Age is not just a rhetorical flourish but a pointer to a specific cascade of molecular events. As the process degrades, it appears to disrupt how proteins are recycled and how energy is delivered to synapses, which in turn leads to those synaptic problems that show up in cognitive testing. The fact that this disruption intensifies with age offers a plausible explanation for why Alzheimer’s risk does not rise gradually but accelerates sharply in later decades.
Protein glitches that tip aging into disease
For years, scientists have focused on amyloid beta plaques as the main villain in Alzheimer’s, but new work suggests that the real damage may start earlier, inside the cell’s protein factories. A report titled Breakthrough Study Reveals How Protein Glitches May Be Driving Alzheimer describes how small errors in protein production and folding can accumulate with age, stressing neurons even before large deposits appear. According to the Feb summary, Breakthrough Study Reveals argues that these glitches may be a driving force that pushes vulnerable neurons over the edge.
The same work points to the possibility that restoring this disrupted process could protect the brain even if plaques are already present. If protein quality control can be reset, neurons might recover some of their ability to clear misfolded proteins and maintain synapses, potentially slowing or even reversing early cognitive decline. That idea reframes Alzheimer’s not only as a disease of toxic buildup but also as a failure of everyday cellular routines that become more fragile with age.
Support cells step into the spotlight
Another piece of the puzzle lies with the brain’s support cells, which far outnumber neurons but rarely get top billing in public discussions of dementia. In Texas, a researcher at Texas A&M has received a federal grant to study how these brain support cells affect Alzheimer risk, a project detailed by Author Michael Mazzuca. The report on a Texas A&M researcher emphasizes that glial cells, which regulate inflammation and help maintain the brain’s environment, may strongly influence who develops Alzheimer and how quickly it progresses.
As these support cells age, they can shift from protective to harmful behavior, releasing inflammatory signals and failing to clear debris efficiently. The Texas project aims to map how those shifts interact with the clogged drains, protein glitches, and synaptic failures described in other studies. If glial cells amplify or dampen those hidden brain processes, therapies that target them could become a way to lower risk even in older adults whose neurons have already begun to struggle.
From early warning to everyday decisions
Taken together, these findings begin to explain why Alzheimer’s risk rises so sharply with age: multiple quiet systems, from perivascular spaces to protein factories to support cells, gradually lose their resilience, and at some tipping point the brain can no longer compensate. The identification of clogged brain drains in Jan, the detailed biochemical links in Enlarged perivascular spaces, the synaptic maintenance problems described by Ivan Farkas, and the protein glitches highlighted in Feb all point to the same conclusion. Age does not simply add years to the clock; it chips away at the machinery that keeps neurons clean, connected, and responsive.
For families and clinicians, the immediate payoff may be in earlier detection and more personalized prevention. If routine imaging can flag clogged perivascular spaces, if blood tests can pick up biochemical signatures tied to Enlarged spaces, and if cognitive screening can catch the subtle synaptic changes described in Hidden Brain Process May, then intervention can start long before a formal diagnosis. Public health resources such as health information hubs and specialized sections on mental health are already beginning to translate these technical advances into guidance on brain wellness across the lifespan, signaling a shift from reacting to Alzheimer’s late to managing brain aging much earlier in life.